Patient population
Twenty-nine consecutive heart transplant recipients referred for exclusion of cardiac allograft vasculopathy and assess allograft function were evaluated with contrast-enhanced retrospectively ECG-gated cardiac DSCT. Additional cardiac MR examination was performed within 1 month before the DSCT examination. Standard exclusion criteria for coronary MDCT angiography and MRI were applied. To be eligible for this study heart transplant recipients had to be in sinus rhythm and present normal renal function (creatinine <1.4 mg/dl). Individuals with known allergy to iodated contrast media, renal failure (creatinine >1.4 mg/dl), arrhythmia (i.e. atrial fibrillation) or frequent extrasystolia (more than four premature beats per minute) were not included. Claustrophobic subjects or patients with MR unsafe ferromagnetic objects, pacemaker or AICD implantation were also excluded. No medication was given to lower or stabilize the heart rate prior to scanning. The study protocol was approved by the institutional review board and all patients gave written informed consent.
DSCT image acquisition, reconstruction, and analysis
Retrospectively ECG-gated cardiac CT exams were performed with a DSCT scanner (Somatom Definition, Siemens Healthcare, Forchheim, Germany) at end inspiration. Scanning direction was craniocaudal initializing above the origin of the coronary arteries and ending at the dome of the diaphragm. The following parameters were employed: 120 kV tube voltage, 410 mAs current with automatic tube current modulation (maximum tube current between 30 and 80% of the cardiac cycle), 32 x 0.6 mm detector collimation, 64 x 0.6 mm slice acquisition, and variable pitch (0.3-0.45) automatically adapted to the heart rate. DSCT studies were acquired after the injection of 70 ml of contrast material (Iomeron 400, Iomeprol, Bracco s.p.a, Milan, Italy) followed by a bolus of 50 ml chaser saline flush at 5 ml/s flow rate using a dual-head power injector (CT Stellant, Medrad Inc. Indianola, USA). Data acquisition was triggered after blood density reached an arbitrarily set threshold value of 100 Hounsfield Units (HU) in the aorta. Mean acquisition time was eight seconds. Image series were directly reconstructed from the raw-data in 5% steps throughout the entire cardiac cycle (0-95% of the R-R interval) in the axial plane with 5 mm section thickness, no interslice gap, medium smooth convolution kernel (B26f), and image matrix of 512 x 512 pixels. No manual ECG-editing was performed. All DSCT images were suitable for analysis. Reconstructed images were transferred to an external workstation (Leonardo, Siemens Healthcare) equipped with a dedicated cardiac post-processing software tool (Argus, Siemens Healthcare).
MRI image acquisition protocol
MRI studies were performed in a 1.5-T MRI system (Magnetom Symphony with quantum gradients; maximum gradient amplitude 30 mT/m; slew rate, 125 mT/m/s, Siemens Healthcare, Erlangen, Germany) using a four-element phased array coil. Retrospectively ECG-gated 7–9 contiguous axial 5 mm slice-thickness cine loops encompassing the entire left atrium were acquired using a steady-state free precession (SSFP) sequence. The following parameters were employed: TR: 3.09 ms, TE: 1.3 ms, flip angle: 80, matrix: 156×192, field of view: 260–280×325–375 mm, twenty-five phases per cardiac cycle. In-plane resolution was 1.7×1.7 mm and temporal resolution varied from 25–50 ms. No interslice spacing was applied. All MRI images were suitable for analysis. Reconstructed images were transferred to the same external workstation.
DSCT and MRI data analysis
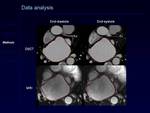
Two independent readers blindly assessed DSCT and MRI exams. In each individual images showing the largest and smallest left atrial cavity areas were visually determined and manually marked as the images representing end-diastolic and end-systolic phases, respectively. On these axial images observers manually traced left atrial boundaries using Argus software. The atrial appendage was included in the volumetric analysis whereas the pulmonary veins were excluded from the calculation (22). Left atrial end-diastolic volume (EDV), end-systolic volume (ESV), and ejection fraction (EF) were estimated for all heart transplant recipients using Simpson’s method (23). Length of time needed for manual contouring was recorded for each imaging technique.
Statistical analysis
Normal distribution of data was assessed with the Shapiro-Wilk test. Data are summarized as mean ± standard deviation. Paired sample Student t test was utilized to estimate left atrial parameter differences for respective DSCT and MRI values. For linear correlation analysis the Pearson correlation coefficient R was calculated defining correlation as poor (r = 0-0.09), minimal (r = 0.1–0.40), moderate (r = 0.41–0.60), good (r = 0.61–0.80) and excellent (r = 0.81–1.0). Bland and Altman plots including mean differences and limits of agreement were generated to ascertain the degree of agreement between DSCT and MRI results for each pair of left atrial parameters (24). Calculation of concordance correlation coefficient (CCC) allowed assessing degree of concordance for each measured variable and estimating interobserver variability (25). The CCC can range from 0 to 1, with CCC of zero representing no agreement and CCC of 1 meaning perfect reliability. A p-value equal or less than 0.05 was considered statistically significant. Data analysis was performed using commercially available statistical software packages (MedCalc, Version 9.3.0.0. MedCalc Software; Mariakerke, Belgium and SPSS for Windows, Version 15.0/SPSS Inc., Chicago, IL, USA).